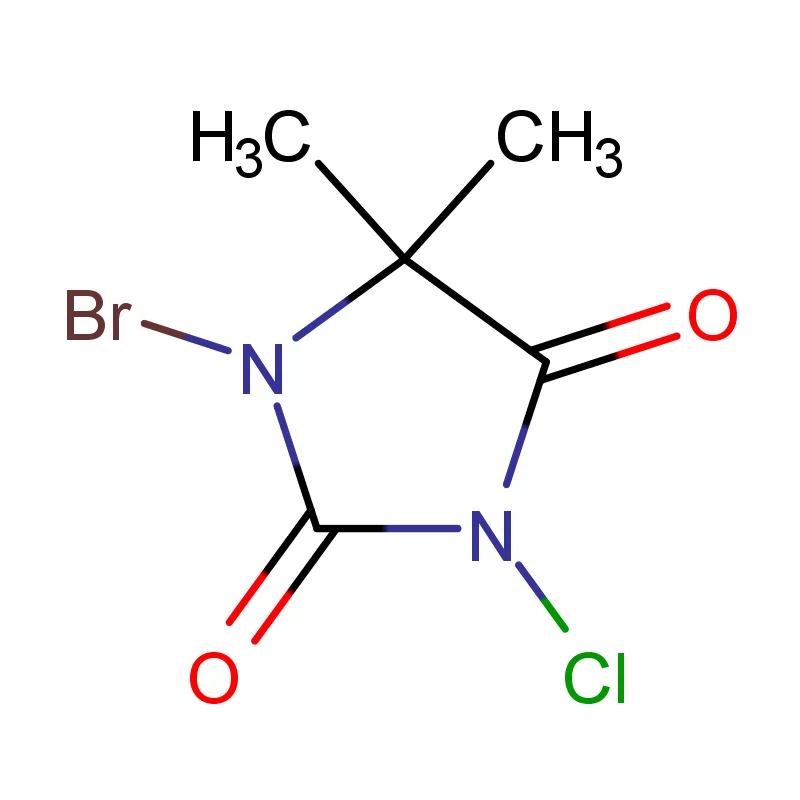
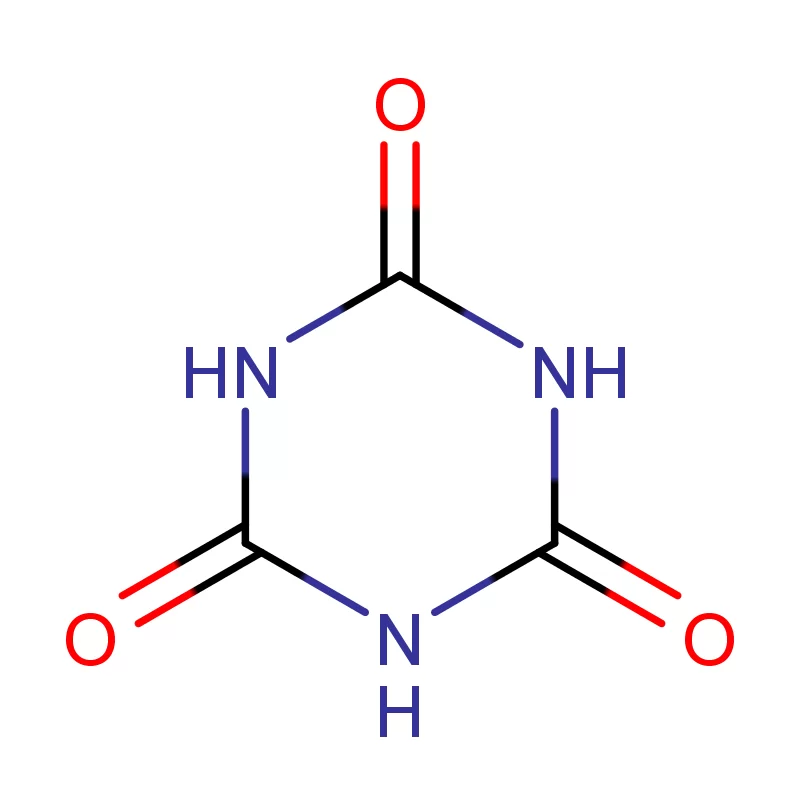
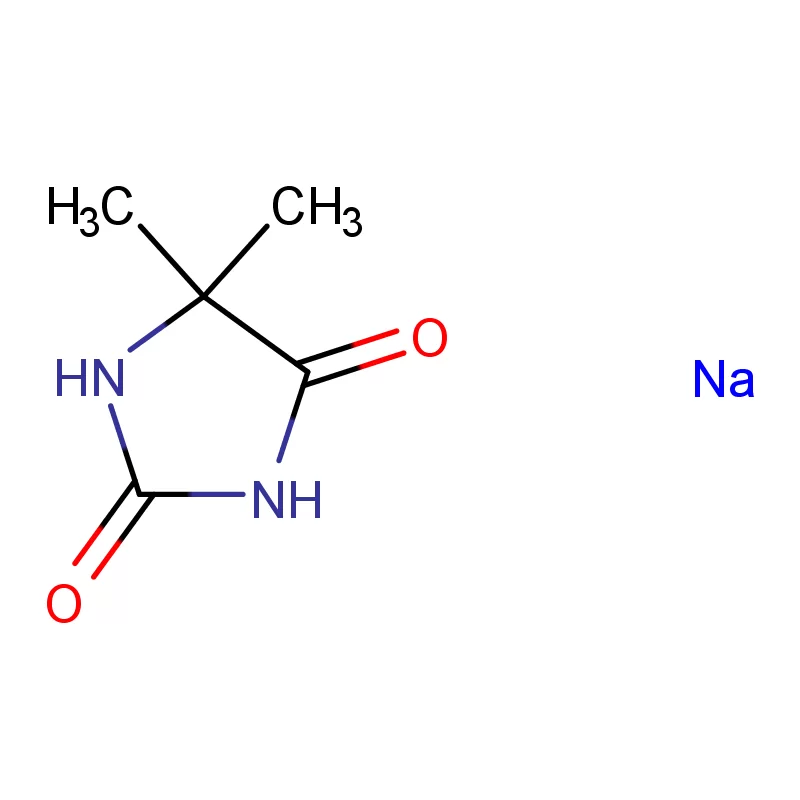
Nigeria, as Africa’s most populous country, presents a significant and growing market for pharmaceutical intermediates. However, the industry faces challenges including reliance on imports, limited local manufacturing capacity, and the need for stricter quality control measures. The tropical climate and prevalence of certain diseases also influence demand for specific pharmaceuticals.
The pharmaceutical industry in Nigeria is characterized by a large number of small and medium-sized enterprises (SMEs) alongside a few larger, more established companies. Access to fine chemicals and consistent supply chains are critical for these businesses. The increasing awareness of healthcare and growing disposable incomes are driving demand for improved pharmaceutical products.
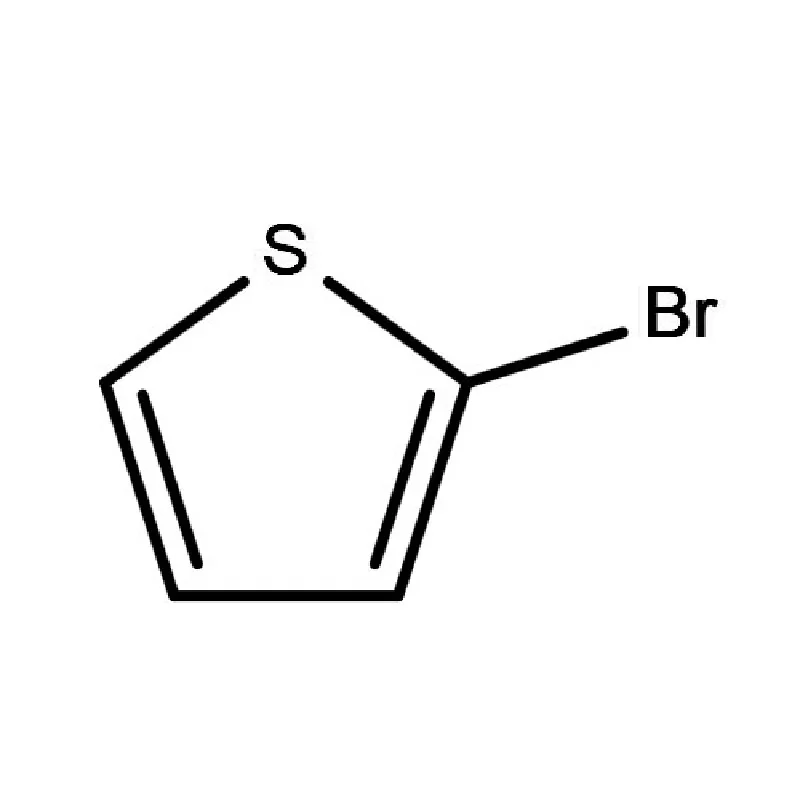
Regulatory hurdles and counterfeit drugs remain significant concerns. The National Agency for Food and Drug Administration and Control (NAFDAC) plays a vital role in ensuring product quality and safety. Demand for 2 Bromothiophene is driven by the production of certain APIs in the region.